
This is to show that the subscript applies to the entire polyatomic ion. For example, iron(II) has a 2+ charge iron(III) a 3+ charge. CID 767 (Carbonic Acid) Component Compounds. Roman numeral notation indicates charge of ion when element commonly forms more than one ion. Computed by PubChem 2.1 (PubChem release 2021.05.07) Parent Compound. Calcium loses two electrons to participate in the ionic bonding with carbonate (an anion) which gives it its +2 charge. 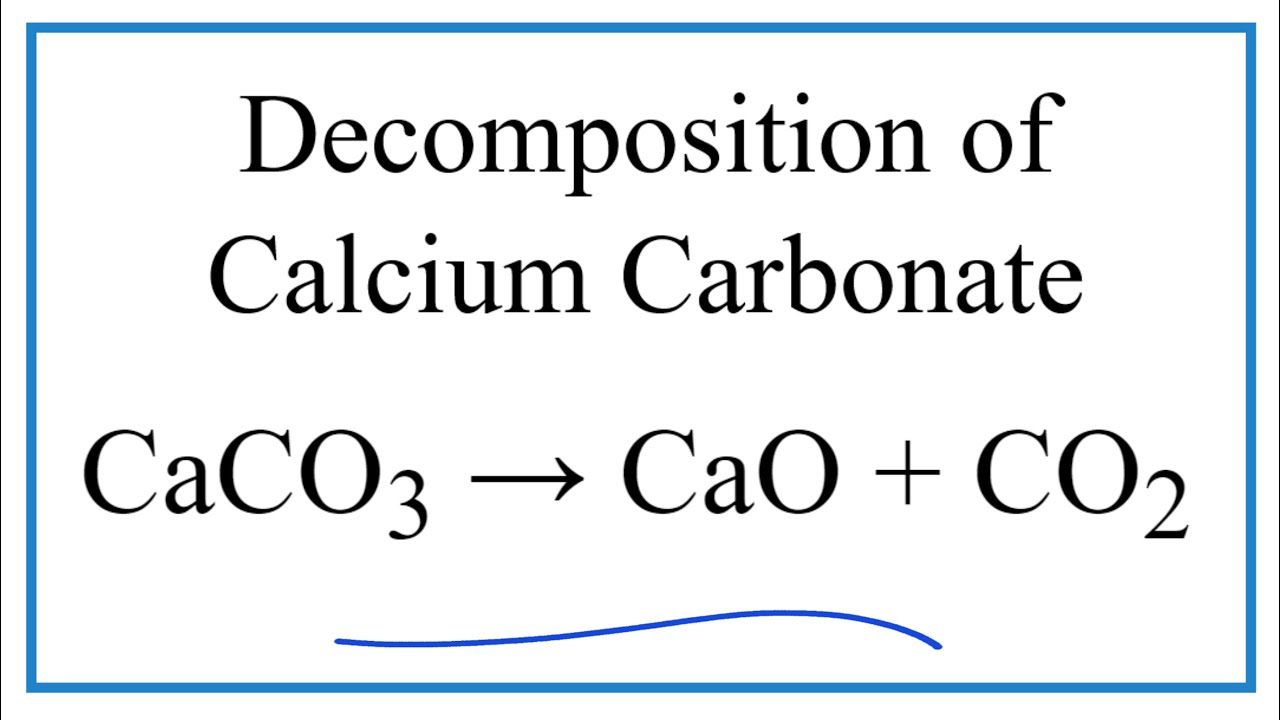
Because the overall compound must be electrically neutral, decide how many of each ion is needed in order for the positive and negative charges to cancel each other out. If more than one of a particular polyatomic ion is needed to balance the charge, the entire formula for the polyatomic ion must be enclosed in parentheses, and the numerical subscript is placed outside the parentheses. Calcium carbonate (calcite) Calcium carbonate (precipitated) Molecular Weight. Start by writing the metal ion with its charge, followed by the nonmetal ion with its charge. In the oxides, when you go from magnesium oxide to calcium oxide, for example, the inter-ionic distance increases from 0.205 nm (0.140 + 0.065) to 0.239 nm (0.140 + 0.099) - an increase of about 17. Because of these opposite charges, these ions naturally pair with one another (similar to two magnets). The oxide ion is relatively small for a negative ion (0.140 nm), whereas the carbonate ion is large (no figure available). Carbonate (CO 3 2-) has a negative charge. Hydrogen carbonates are amphoteric because they act as both weak acids and weak bases. (credit: Mark Ott) Other applications of carbonates include glass makingwhere carbonate ions serve as a source of oxide ionsand synthesis of oxides. The rule for constructing formulas for ionic compounds containing polyatomic ions is the same as for formulas containing monatomic (single-atom) ions: the positive and negative charges must balance. When we talk about ionic imbalance and its relationship to pH, we are talking about the concentration of magnesium, calcium, boron, and carbonate ions. 2: The reaction of calcium carbonate with hydrochloric acid is shown.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
